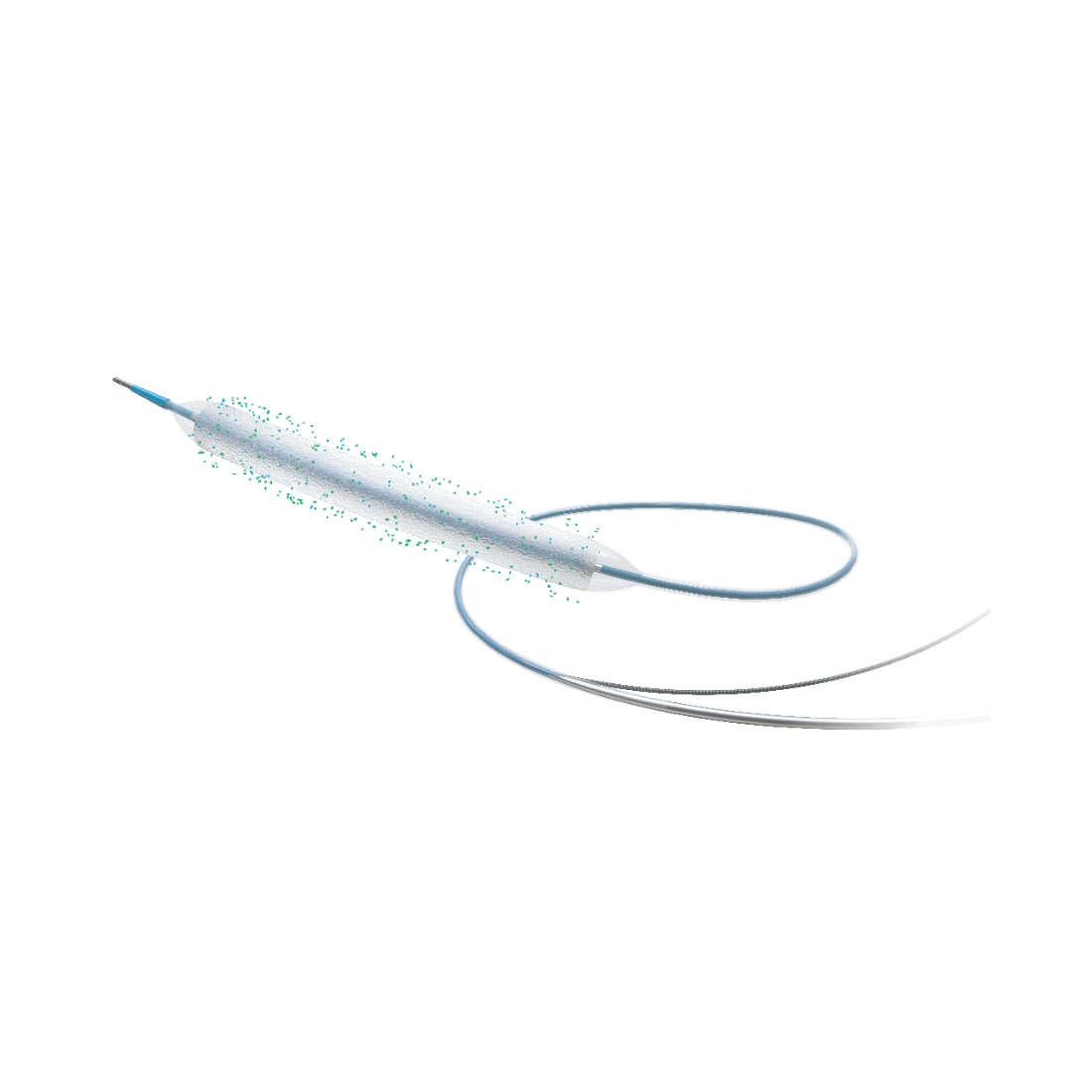
Superior Efficacy, Reduced Restenosis
• Utilizes the proven antiproliferative agent paclitaxel, whose lipophilic properties enable efficient tissue absorption.
• Incorporates an innovative maltose-based excipient for stable drug loading and effective release, providing sustained therapeutic action for up to 28 days.

Innovative Choice, Implant-Free Intervention
• More suitable for delicate intracranial vessels.
• Reduced postoperative management burden, with no requirement for long-term dual antiplatelet therapy.
• Avoids chronic inflammatory reactions associated with metal.
Conformable and Smooth Delivery
• Advanced thin and uniform coating technology effectively reduces the balloon outer diameter while maintaining adequate drug dosage and improving balloon compliance.
• Rapid-exchange delivery system with ultra-smooth distal coating enhances deliverability.
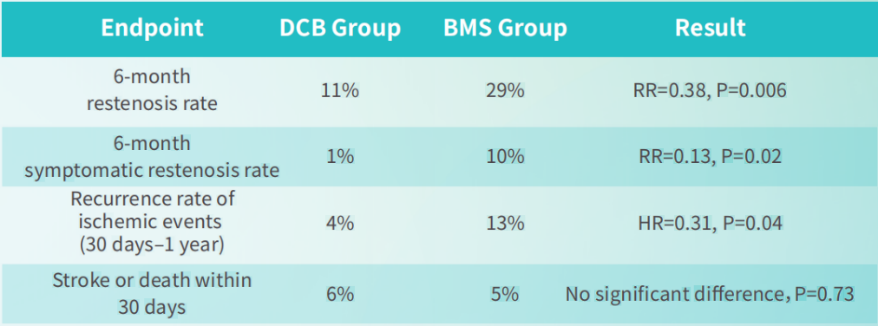
Robust Clinical Evidence, Proven Safety and Effectiveness
The DR.BEYOND clinical study demonstrated that in the treatment of symptomatic intracranial atherosclerotic stenosis (sICAS), compared with conventional bare metal stents (BMS), CQ18™ drug-coated balloon (DCB) significantly reduced the risk of restenosis and recurrent ischemic events without increasing early safety risks.
The results were published in the leading radiology journal Radiology.
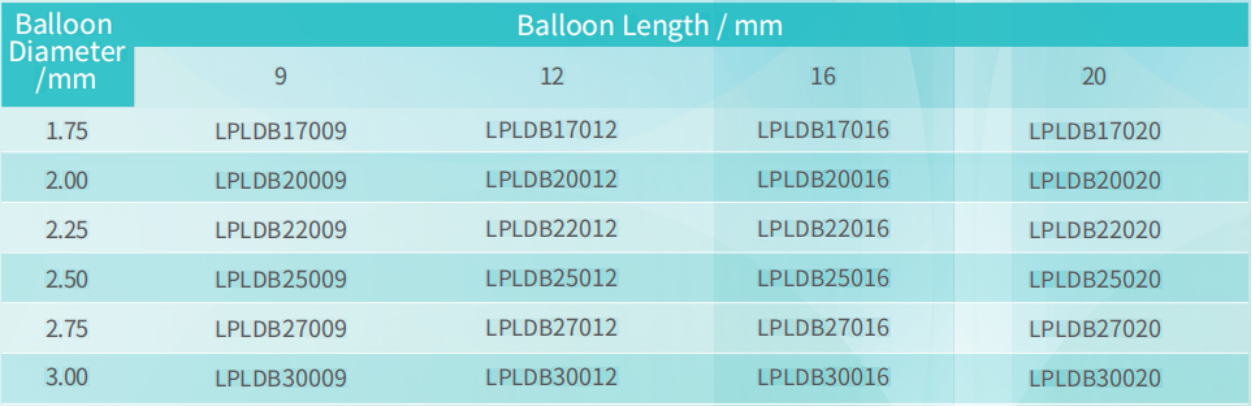
Product Specification
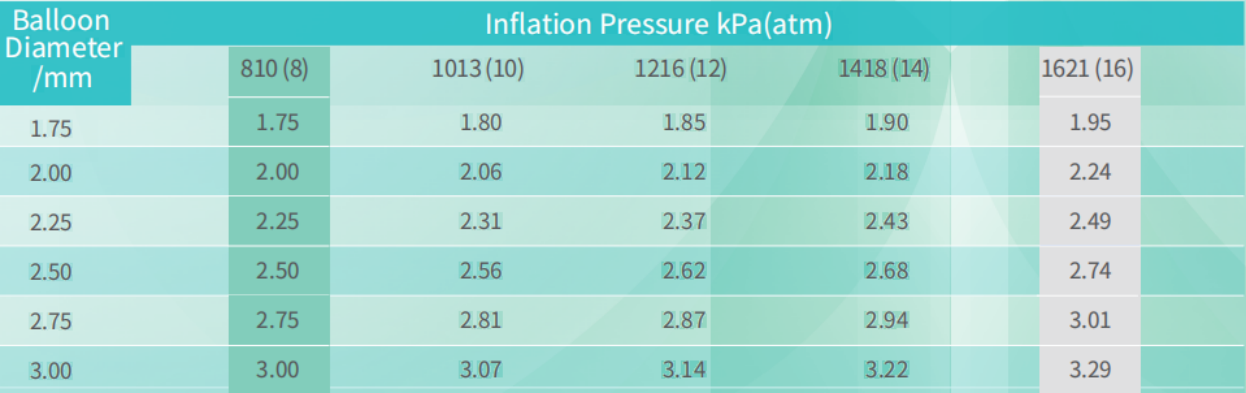
Relationship Between Balloon Diameter and Inflation Pressure

* Product registration and availability vary by country. For more information on product availability, please contact us.
Thank You for Your Attention on TJWY Medical!
Email us with any questions or inquiries or use our contact data. We would be happy to answer your questions.